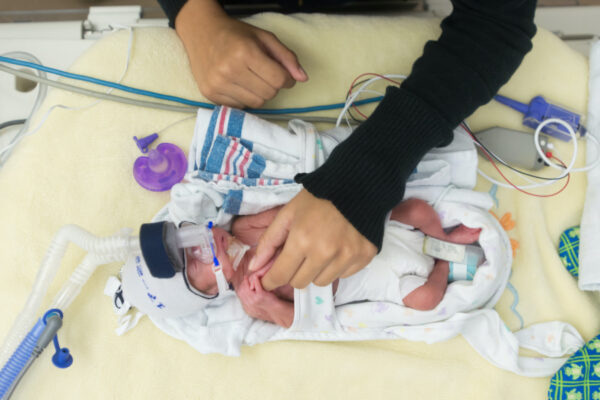
Humans are in motion long before we’re born: In utero, fetal movements promote the healthy development of vital organs, the nervous system and more.
And when babies are born, studying their movement patterns presents a critical opportunity. Certain patterns can predict when preterm infants — those born before 37 weeks of gestation — will experience one of several common, yet dangerous, events like pauses in breathing (apnea) or a slowed heart rate (bradycardia). Beyond a stay in the neonatal intensive care unit, these events can indicate babies’ long-term neurological health well into adulthood, so the ability to predict — and, ultimately, prevent — them could fundamentally change neonatal care delivery.
These findings and more are all part of a large-scale data collection and analysis effort led by David Paydarfar, M.D., chair of Dell Med’s Department of Neurology, and Ally Richardson, a graduate student at theOden Institute for Computational Engineering and Sciences. Funded by a grant from the National Science Foundation, their team and collaborators at Dell Children’s Medical Center of Central Texas are monitoring the vital statistics of babies in the NICU, aiming to understand — and, eventually, reduce — the complexity of care for these vulnerable patients.
“We’re trying to make the case that movement is a vital sign, just like blood pressure, pulse, respiration or temperature,” Paydarfar says. “We know that preterm infants are at risk of many neurological complications later in life, like cerebral palsy, autism spectrum disorder or learning disabilities, and tracking on some of these early events can help us better understand the relationship to those later outcomes more clearly.”
From Measurement to Intervention
The team is using data already collected in the NICU to study more than 100 babies’ statistics. Rather than rely on video monitors or accelerometers, which are sensitive to failure or get in the way of basic care, the team developed an algorithm to accurately track movement patterns via blood-oxygen saturation levels, which are already collected as part of routine care.
The results can be, at times, counterintuitive.
“If I get up and I start running, I breathe more,” Paydarfar says. “Well, in preterm infants who have very immature movement patterns, they are more likely to do the opposite. They stop breathing — their heart rate goes down. So we see a direct, paradoxical effect in these bursts of movement, and we can actually use movement to predict when the next apnea is going to be.”
The team uses machine-learning algorithms to predict the adverse events, which could one day lead to tools like a smart mattress that gently vibrates an infant when it detects that an apnea may occur, or monitors that signal to hospital staff when a baby is about to experience bradycardia.
Producing effective clinical tools from the raw biological data, though, means taking a brand-new computational approach.
“In these high-stakes decision-making situations, clinicians need to be able to verify why the machine-learning model is classifying an apnea or other event, and most black-box algorithms don’t do that,” Richardson says. “Something I’m really passionate about is creating an algorithm and a tool that openly gives doctors all the information needed to verify so that they can trust the data and make good decisions in those moments.”
The ‘Holy Grail’ of Predictions
For John Loyd, M.D., associate professor in Dell Med’s Department of Pediatrics and chief of the Division of Neonatology at Dell Children’s Medical Center, the opportunities for the modeling effort to impact care are stark, and go beyond responding to life-threatening events.
“The holy grail of this would be a clear indicator that something bad is about to happen with a baby, which we could then respond to before it happens,” Loyd says. “But the second thing is also to be able to more clearly identify those who are safe for discharge. With movement, we may be able to more clearly identify infants that are developmentally ready for discharge.
“A project like this shows us how a medical school can benefit our local community, as our system of care continues to get more sophisticated thanks to these kinds of research partnerships.”
The NSF-funded research grant entitled “Movement as a Vital Sign in Preterm Infants” has been conducted in collaboration with Dagmar Sternad, Ian Zuzarte and Leonora Blodgett at Northeastern University, and Premananda Indic at The University of Texas at Tyler.